Technology
13-M is Serca Pharmaceutical’s novel drug candidate in preclinical development for treatment of acute myocardial infarction. The clinical aim is to provide a medical treatment to patients with acute myocardial infarction who are subject/eligible for PCI procedure – either immediate after hospitalization (patients with MI that show ST elevation upon EKG examination (STEMI patients)) or PCI within 24-48 hours after initial hospitalization (non-STEMI patients).
13-M is a small molecule shown to have important protein to protein interactions (PPI) characteristics with the capacity to modulate the Ca2+ pump, i.e. the SERCA2 in the (figure 1).
Specifically, 13-M targets the AKAP18d-PLB interaction and thereby interrupting the activation of SERCA2 (figure 2) and thereby the contractile force and energy consumption upon adrenergic stress is reduced (figure 2).
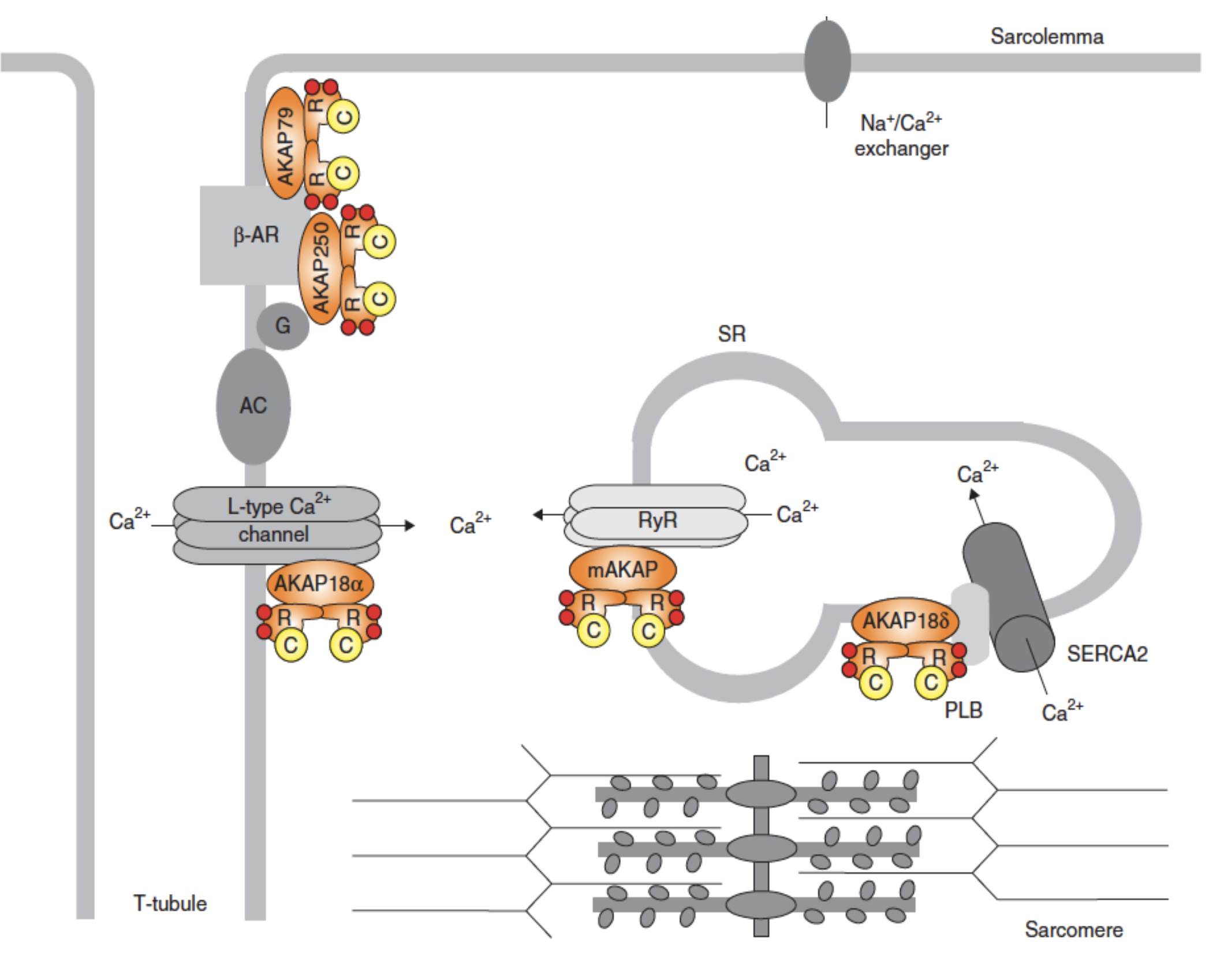
Figure 1: Schematic illustration of a cardiac myocyte and Ca2+ handling. Major components in the excitation–contraction coupling in the myocyte are shown. The cycling of Ca2+ is indicated by arrows and the different protein kinase A (PKA)–A-kinase anchoring protein (AKAP) complexes that provide adrenergic regulation of various components of the Ca2+ handling machinery are indicated. AC: Adenylyl cyclase; β-AR: β-adrenergic receptor; C: Catalytic subunit of PKA; mAKAP: Muscle-specific AKAP; PLB: Phospholamban; R: Regulatory subunit of PKA; RyR: Ryanodine receptor; SERCA: Sarcoplasmic reticulum Ca2+-ATPase; SR: Sarcoplasmic reticulum; Red circle: cAMP.
The mechanism of action by which 13-M exerts its effect is highly selective; it provides its cardioprotective effect by inhibiting the adrenergic contractility (inotropic) of the heart, without affecting the heart rate (chronotropic). The preventive effect on contractility, and thus energy demand, makes 13-M well suited for limiting the myocardial infarction size in patients being treated by percutaneous coronary intervention (PCI; “stenting”). PCI is associated with adrenergic stress and ischemic reperfusion injury (IRI) when the myocardium is re-perfused.
Figure 2: Protein kinase A (PKA)-A-kinase anchoring protein (AKAP)18d–phospholamban (PLB)–sarcoplasmic reticulum Ca2+-ATPase (SERCA)2 complex.
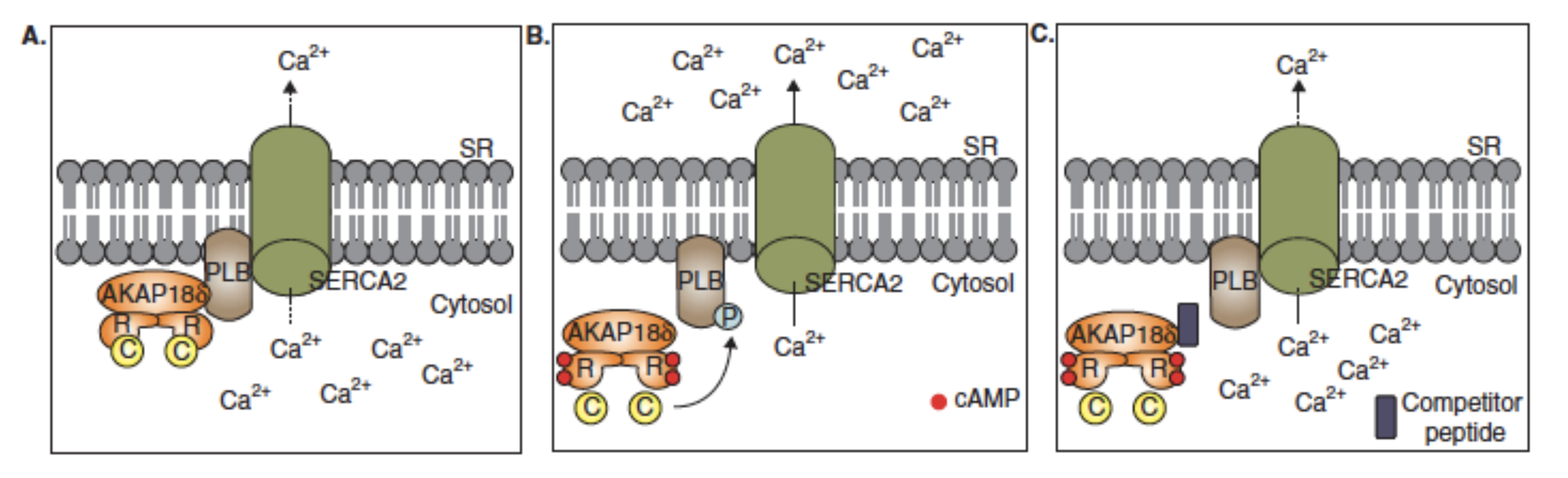
A. Resting situation, no adrenergic drive, low heart rate, SERCA2 inhibited by PLB with low ATP- and energy consumption and low contractility.
B. Adrenergic stimulation paces the heart, increased heart rate. SERCA2 released from PLB inhibition by PKA phosphorylation. Fast Ca 2+ reabsorption, high ATP- and energy consumption and increased contractility.
C. Disruption of AKAP18δ –PLB complex prevents PKA phosphorylation and dissociation of PLB and thereby SERCA2 activation. Low ATP- and energy consumption and a negative inotropic effect. C: Catalytic subunit of PKA; R: Regulatory subunit of PKA; SR: Sarcoplasmic reticulum.
13-M preclinical efficacy
In a rat model, 13-M reduced myocardial infarction size assessed by infarction biomarker troponin (figure 3a) and infarction size (figure 3b). Cardiac troponin (cTpl) is a protein found in cardiac muscle fibers that regulate muscular contraction and is commonly used to help detect heart injury and shown to correlate with final infarct size.
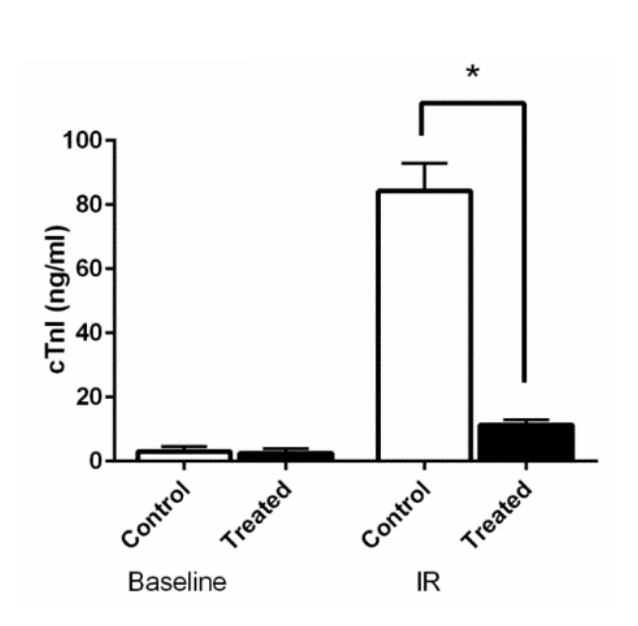
Fig 3a. 13-M treatment significantly decreased the level of cardiac troponin (cTnl)
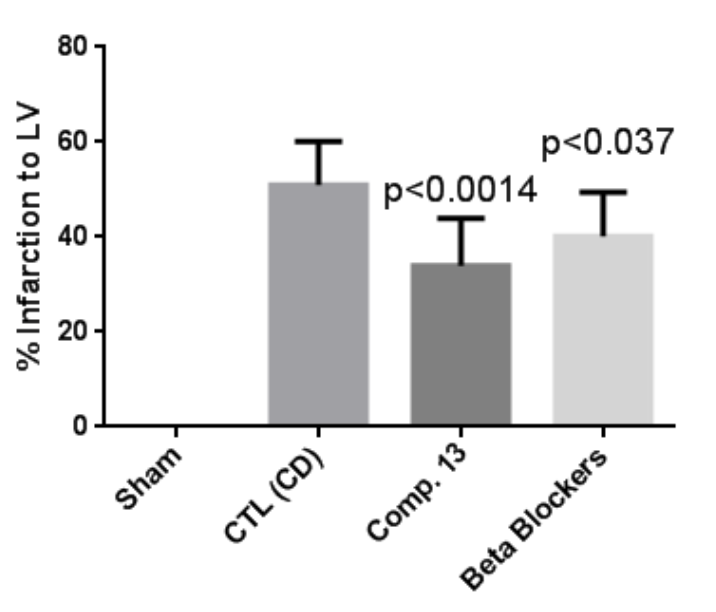
Fig 3b. Final infarct size determined by late-gadolinium -enhancement MRI in rats. Treatment with 13-M showed about 30% decrease in final infarct size.
Heart function was assessed by ultrasound echocardiography after treatment with 13-M in rats (fig 4a,b) and showed that 13-M improves the heart function compared to untreated controls.
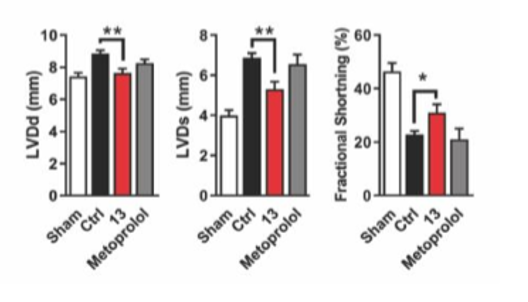
Figure 4a. Graphs show difference in left ventricular diameter in diastole (LVDd) and systole (LVDs) and fractional shortening in the groups.
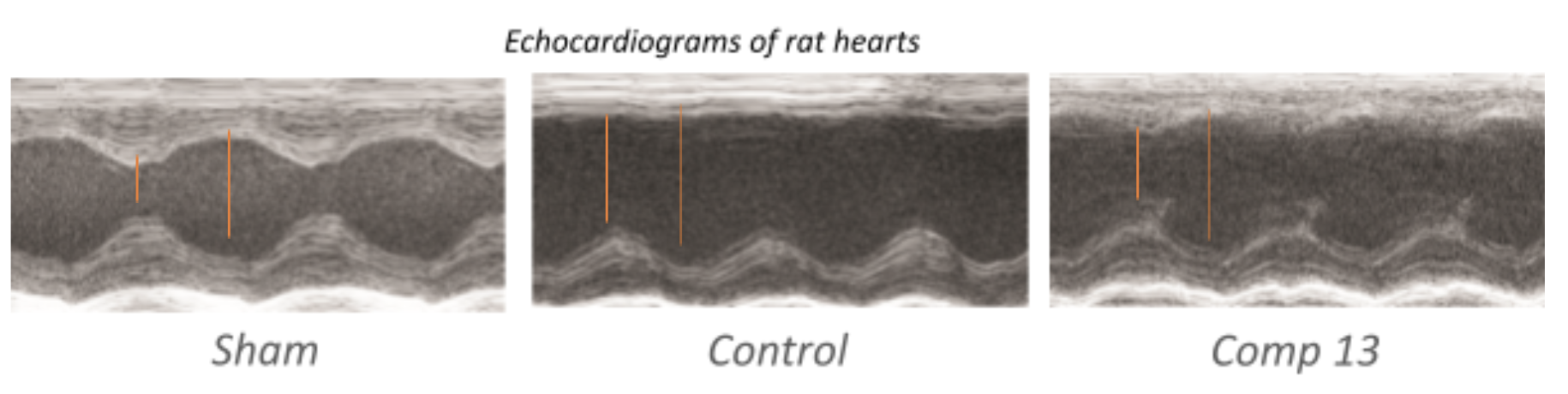
Figure 4b. Representative M-mode echocardiograms 9 days after sham operated animals compared to animals treated with vehicle (control) or 13-M before reperfusion. Red bars outline the left ventricular diameter in diastole (LVDd) and systole (LVDs).
13-M is at Preclincal Product Development stage
13-M is in preclinical development stage. Pharmaceutical product development and preclinical safety, pharmacokinetics and ADME is ongoing at Cadila Pharmaceuticals. Pharmacology studies in animal models is initiated at Contract Research Organizations (CRO) managed by Serca Pharmaceuticals in Europe.


